Microwave Digestion Systems
Microwave acid digestion, microwave acid leaching, and much more
Superior trace elemental analysis begins with outstanding sample preparation. With over 40 years of experience, we’ve made microwave digestion easier and more convenient than ever before – thanks to sophisticated instrument and vessel concepts, state-of-the-art sensor technology, and the highest safety standards. We have a broad range of instruments for all kinds of applications: microwave digestion systems Multiwave 7101/7301/7501, Multiwave GO Plus, the microwave reaction platform Multiwave 5000, and the conventionally heated Multicube 48.
![]() A Chemist’s Guide to Sample Preparation
A Chemist’s Guide to Sample Preparation
Order now for free >>
Anton Paar Products
Multiwave 7101/7301/7501: Demanding samples, easy digestion
With its Pressurized Digestion Cavity (PDC), the Multiwave 7101/7301/7501 series provides an unmatched, streamlined acid digestion workflow that covers your whole range of samples, from routine to high-end applications, in the same run, with one method. You can use these instruments with high (≤4 g) and low sample amounts as well as high (≤60 mL) and low volumes in glass (disposable), quartz, or PTFE vials with plug-on caps.
Multiwave GO Plus - The smallest microwave digestion system
Multiwave GO Plus is the smallest parallel microwave digestion system on the market and enables fast and easy processing of routine applications of all kinds of samples including food and biological samples, EPA procedures, environmental samples, agricultural samples, cosmetic and pharmaceutical samples. Digesting up to 12 samples simultaneously within a compact footprint is unrivalled on the market. Multiwave GO Plus has been designed for the easiest handling with low investment and running costs.
Multiwave 5000 – One system, endless possibilities
Thanks to the flexible platform concept you can configure Multiwave 5000 to be the best fit for your applications. Multiwave 5000 provides: digestion of all kinds of samples (varying in difficulty or volume), acid leaching, microwave solvent extraction, evaporation, microwave-induced oxygen combustion, UV digestion, sample drying, and synthesis. As the user-friendliest microwave system ever created, it saves labs time and money with features such as an easy-to-navigate, smartphone-like user interface, a method library with more than 500 pre-installed programs, SmartScreen, and tool-free easy vessel handling.
Multicube 48 – Trendsetting open-vessel digestion
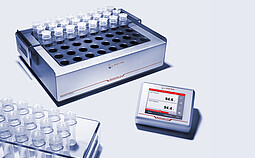
Multicube 48 is a robust laboratory hot block for acid digestion, evaporation, and concentration of samples as well as other applications requiring elevated temperatures. The outstanding design enables high temperature homogeneity of ±1 °C over the whole block to ensure the same digestion quality for all samples. The high-purity, low-cost disposable vials have the lowest metal blanks to eliminate the risk of external contamination of your samples.
The largest global service network of all microwave suppliers available for your support
Anton Paar provides:
- Dedicated local application support specialists throughout the world
- On-site and local service options
- Demonstration laboratories throughout the world and on-site demonstrations available
- Different installation and user training levels are available
Webinars
We offer you a great and constantly growing choice of live webinars and recordings on products, applications, and scientific topics.
View webinars